Foreign products may also have the same code as an FDA approved imprint. For example, an imprint code might be reused after a drug product has been discontinued. There may be circumstances where two products have the same imprint code. that is marked as homeopathic is being marketed without FDA evaluation for safety or effectiveness. There are no FDA-approved homeopathic products. Homeopathic drug products are required only to bear an imprint that identifies the manufacturer and their homeopathic nature.Inclusion of a letter or number in the imprint, while not required, is encouraged as a more effective means of identification than a manufacturer's symbol or logo by itself. Identification of the drug product requires verification of its active ingredients and its dosage strength.The drug manufacturer determines the imprint for specific drug products. An imprint is defined as any single letter, number, or combination of letters and numbers, including words, company names, National Drug Code, or a mark, symbol, logo or monogram.The purpose of regulating imprinting is to enable identification of the pill ingredients and drug manufacturer by law enforcement officials, health care providers and the patient or caregiver. The imprint code, when used in conjunction with the product's size, shape, and color, permits the identification of the drug product, the active ingredient(s), strength, and the manufacturer or distributor of the product. 
you need to double check a medication from the pharmacy that looks different and may be a generic substitution.you have dropped it or mixed it up in a pill box or case. 
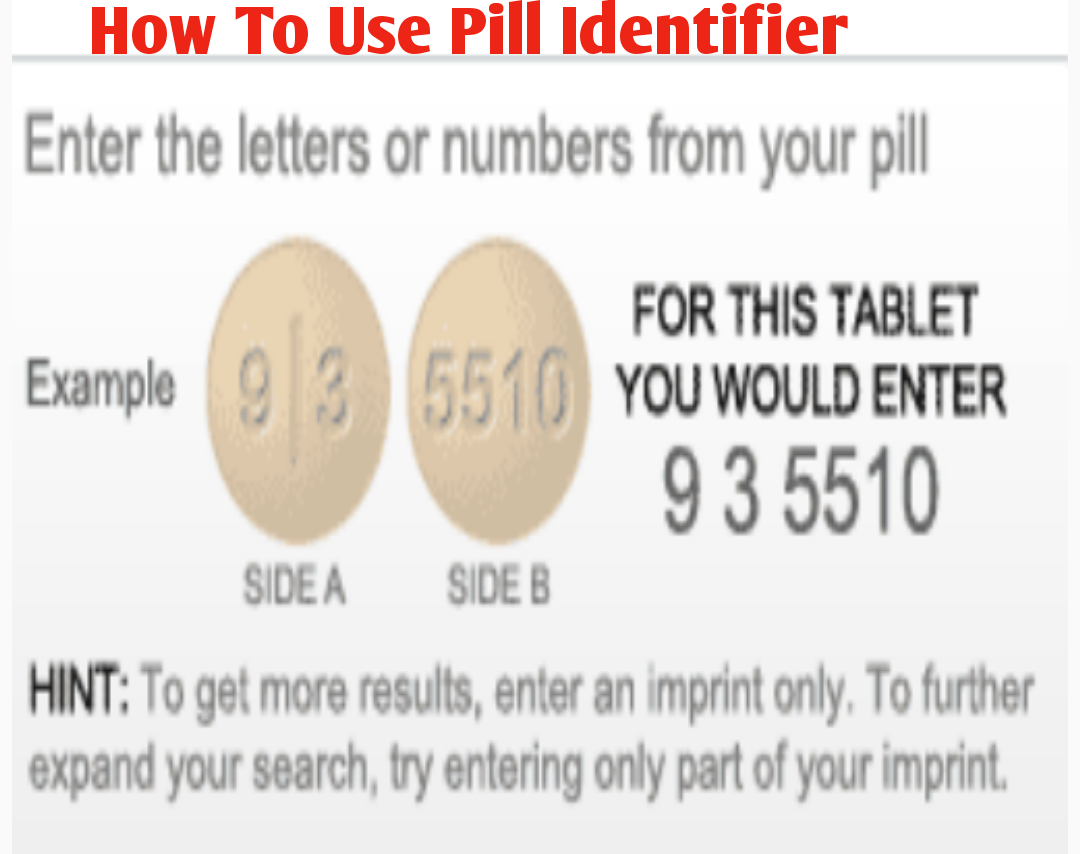
Solid oral dosage forms are defined as capsules, tablets, or similar drug products intended for oral use. This also applies to biological drug products and homeopathic drug products, unless otherwise exempted in the FDA Code of Federal Regulations 206.7. are required by the Food and Drug Administration (FDA) to have an imprint. Need to identify an imprint code? Try the Pill Identifier What are Imprint Codes?Īll approved prescription and over-the-counter solid, oral dosage form medications in the U.S. Medically reviewed by Leigh Ann Anderson, PharmD.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
