|
Specific challenges for SCS remain, especially for pain conditions such as CRPS I and causalgia that differ by etiology and symptom profile from other chronic pain syndromes. 26 Spinal cord stimulation (SCS), for which electrodes are placed into the dorsal epidural space, is an available treatment of a variety of chronic neuropathic pain conditions such as failed back surgery syndrome and complex regional pain syndrome (CRPS). The prevalence of neuropathic pain refractory to the current standard of care has been estimated to be 1.5% of the general population. As the largest prospective, randomized comparative effectiveness trial to date, the results show that DRG stimulation provided a higher rate of treatment success with less postural variation in paresthesia intensity compared to SCS. Finally, subjects using DRG stimulation reported less postural variation in paresthesia ( P < 0.001) and reduced extraneous stimulation in nonpainful areas ( P = 0.014), indicating DRG stimulation provided more targeted therapy to painful parts of the lower extremities. Dorsal root ganglion stimulation also demonstrated greater improvements in quality of life and psychological disposition. Device-related and serious adverse events were not different between the 2 groups. The percentage of subjects receiving ≥50% pain relief and treatment success was greater in the DRG arm (81.2%) than in the SCS arm (55.7%, P < 0.001) at 3 months. No subjects reported stimulation-related neurological deficits. The predefined primary composite end point of treatment success was met for subjects with a permanent implant who reported 50% or greater decrease in visual analog scale score from preimplant baseline and who did not report any stimulation-related neurological deficits. The primary end point was a composite of safety and efficacy at 3 months, and subjects were assessed through 12 months for long-term outcomes and adverse events. Subjects received neurostimulation of the DRG or dorsal column (spinal cord stimulation, SCS). ACCURATE, a pivotal, prospective, multicenter, randomized comparative effectiveness trial, was conducted in 152 subjects diagnosed with complex regional pain syndrome or causalgia in the lower extremities. JUDE MEDICAL APPROVED TRIAL LEADS AND EXTENSIONS TO DELIVER TRIAL STIMULATION FOR A MAXIMUM OF 30 DAYS.Animal and human studies indicate that electrical stimulation of dorsal root ganglion (DRG) neurons may modulate neuropathic pain signals. JUDE MEDICAL INVISIBLE NEUROMODULATION TRIAL SYSTEM IS INTENDED TO BE USED EXTERNALLY WITH ST. JUDE MEDICAL INVISIBLE TRIAL SYSTEM AND IS INDICATED AS AN AID IN THE MANAGEMENT OF CHRONIC, INTRACTABLE PAIN OF THE TRUNK AND/OR LIMBS, INCLUDING UNILATERAL OR BILATERAL PAIN ASSOCIATED WITH ANY OF THE FOLLOWING: FAILED BACK SURGERY SYNDROME AND INTRACTABLE LOW BACK AND LEG PAIN. JUDE MEDICAL PATIENT CONTROLLER APP (PC): MODEL 3871 AND 4) ASSOCIATED ACCESSORIES (LEAD MODELS 3046, 3066, AND 3086 AND MULTI-LEAD TRIAL CABLE (MLTC) 3013).THE DEVICE, AS MODIFIED, WILL BE MARKETED UNDER THE TRADE NAME ST. JUDE MEDICAL CLINICIAN PROGRAMMER APP (CP): MODEL 3870 3) ST. JUDE MEDICAL EXTERNAL PULSE GENERATOR (EPG), 16 CHANNEL: MODEL 3599 2) ST. JUDE MEDICAL INVISIBLE TRIAL SYSTEM COMPRISING OF THE FOLLOWING DEVICES:1) ST. Stimulator, spinal-cord, totally implanted for pain reliefĬhange Design/Components/Specifications/MaterialĪPPROVAL FOR THE ST. GENESIS AND EON FAMILY SPINAL CORD STIMULATION (SCS) SYSTEM Be sure to look at the original PMA record for more information. A supplement may have changed the device description/function or indication from that approved in the original PMA.  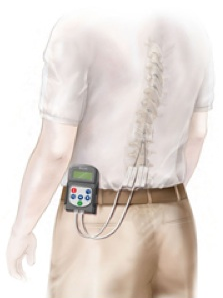
Note: This medical device record is a PMA supplement.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
